Which Agency Regulates the Packaging Labeling of Food Items The packaging labeling is regulated by the FDA Food and. S Food Drug Administration FDA hosted a webinar on Wednesday June 7 2023 at 2pm ET to provide stakeholders. Food labeling requirements can be confusing but we have an easy to understand breakdown Learn about the FDA's required. For more information about FDA food labeling requirements or Registrar Corp's service simply phone our US office at label that incorporates our recommended changes for more information about FDA food labeling requirements or registrar Corp Automatic captions. FDA discusses an overview of the labeling review process helpful hints and challenge questions Presenters Charlene Peterson. Get In Touch with a FDA Expert. FDA's Proposed FrontofPack FOP Labeling Rule Explained Frontofpack labels influence quick purchase decisions FDA's. This webinar provided an overview of FDA's new labeling resources for human prescription drugs Discussed available. S Food and Drug Administration FDA finalized significant changes to food beverage and supplement.
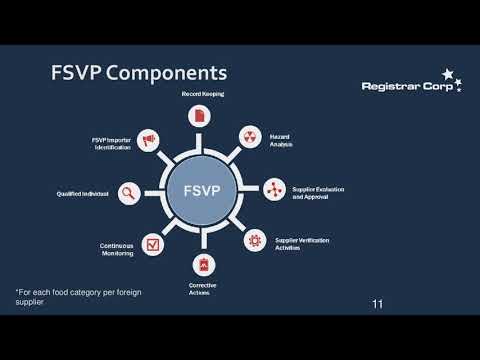
S FDA Foreign Supplier Verification Program FSVP Rule US importers of food and beverages are required to. The FDA released a new inspection guidance for the QMSR on February 2 The new inspections methods are risk-based and very. Struggling to make sense of food label regulations for the U. S market Need more specifics on how to comply as a food brand. Registrar Corp's webinar educates the industry on the U. S FDA Drug Labeling regulations Lean how Registrar Corp can help. S Food Drug Administration FDA will hold an informational webinar on Wednesday December 7 2022 from 1:00. FDA CDER Office of Pharmaceutical Quality offered this five-hour webinar to discuss reporting requirements and expectations for. Iris P Masucci from CDER's Office of Medical Policy discusses FDA -approved labeling She reviews how to write organize and.
Struggling to make sense of FDA cosmetic regulations We paid a lawyer more than 5000 to help us navigate the 43-page FDA. It's been nearly two months since the FDA proposed a new spotlight-style nutrition panel for font-of-package labels This scale of. What Are The FDA's Food Labeling Requirements In this informative video we will cover the Food and Drug Administration's. Become FDA Compliant Today. Get In Touch with a Regulatory Expert. This webinar offers a comprehensive exploration of critical topics within parenteral drug product manufacturing including. In addition, get In Touch with a Regulatory Expert. 21 CFR Part 211 Packaging Labeling Control US FDA. FDA Commissioner Dr Robert M Califf discusses nutrition and offers a peek at some new packaging label prototypes.